We are updating our documentation. The new Lymed product catalogue already has the new content, but we are also updating the instructions for use (IFU), order forms, packaging labels, and the website to match.
Download the new Lymed product catalogue here
These updates are part of our continuous improvement efforts and the harmonization of our documentation. At the same time, we are developing our materials to more clearly support consistent practices, compliance, and traceability.
Here are the highlights of this process
INSTRUCTIONS FOR USE
The new Lymed Instructions For Use (IFU) is available for download now. The printed IFU will start shipping with every Lymed product soon.
The structure and legibility have been improved in the new version. The wording about using, caring for, and donning and doffing of the product have all been made clearer. The intended use or method of use of the product is not affected by these content updates.
We have also added new languages to better serve our customers.
ORDER FORMS
The Lymed order forms are the medium we use to create fully individualized medical pressure garments for your clients! This is why it is so important to fill it out carefully. Please make sure to download and start using the new forms right away!
Download the new order forms here
New information fields on pages 1 and 2:
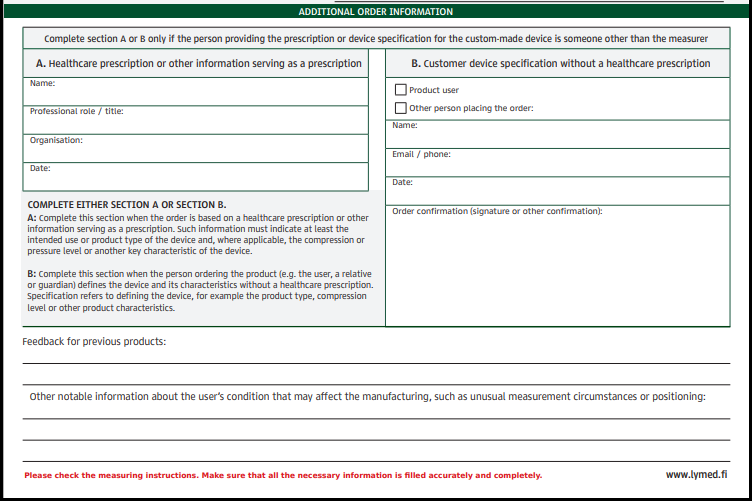
We will collect more detailed information about the person providing the prescription or device specification for the Lymed custom-made device. Specification refers to defining the device, for example the intended use, product type and compression/pressure level.
The new fields must be completed if the measurer isn’t person providing the prescription or device specification.
The information of the person specifying the device is filled out in the information fields:
- when the order is based on a healthcare prescription or other information serving as a prescription.
- when the person ordering the product (e.g. the user, a relative or guardian) defines the device and its characteristics without a healthcare prescription.
If the measurer makes the order and specifies the device features, then the extra information fields need not be filled out.
Updates on the measurement pages
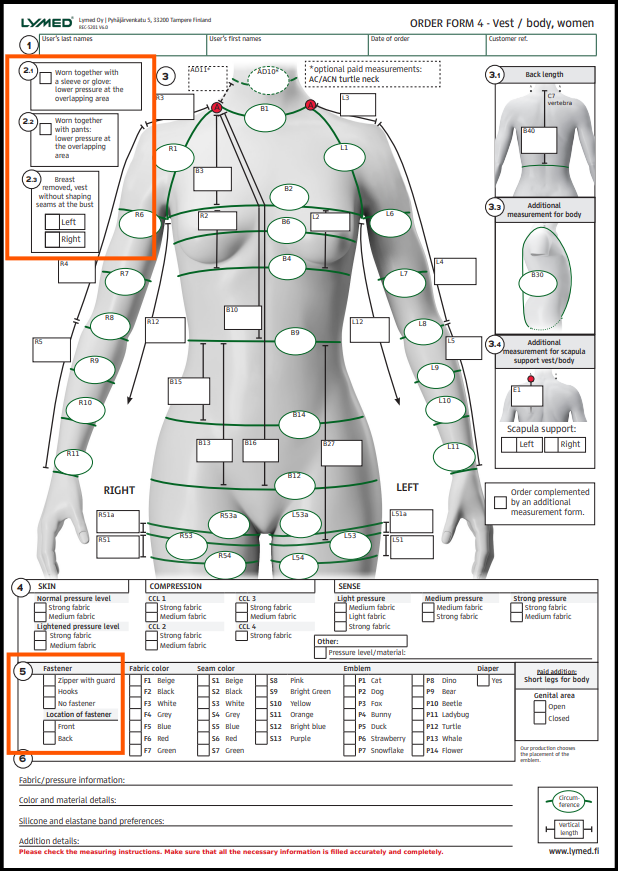
On several pages the additional fields 2.1, 2.2, and 2.3 now have clearer instructions.
Field 5 has been updated for clarity: there is no longer a default closure method and instead hooks or a zipper (etc.) must be marked when placing the order.
PACKAGING LABELS
The packaging labels have seen an improvement in their content as well as the graphic design. We have added elements to make it easier to tell apart different kinds of products and to make the product information clearer.
For standard-sized products there’s a transition period, and old labels may be shipped while supply lasts.
Do you have questions? You can contact us on email at info@lymed.fi or call during office hours (Finnish time, EET/EEST) +358 20 7792233.